Gujarat Board GSEB Textbook Solutions Class 11 Chemistry Chapter 11 The p-Block Elements Textbook Questions and Answers.
Gujarat Board Textbook Solutions Class 11 Chemistry Chapter 11 The p-Block Elements
GSEB Class 11 Chemistry The p-Block ElementsText Book Questions and Answers
![]()
Question 1.
Discuss the pattern of variation of oxidation states in
(i) B to Tl
(ii) C to Pb.
Answer:
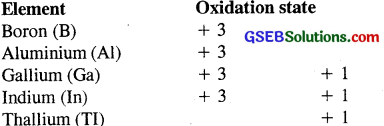
The electronic config. of all the above elements is ns²np². As we go down the group the 2 electrons present in s-orbital prefer to remain paired, i.e., they do not take part in bond formation. Such a pair of electrons is called INERT-PAIR and this effect is called INERT- PAIR EFFECT.
Therefore as we go down the group 13, the + 1.
Oxidation state becomes increasingly more dominant.
(ii)

It is again due to inert-pair effect.
As we go down the group ns² electrons prefer to remain paired, i.e., they do not take part in bond formation. Therefore the heavier elements of the group show increasingly + 2 oxidation state.
Question 2.
How can you explain higher stability of BCl3 as compared to TICl3?
Answer:
BCl3 is more stable than TICl3 because inert pair is missing in BCl3. Due to inert pair effect thallium will prefer to form TICl rather than TICl3. It prefers to show an oxidation state of + 1.
Silicones being surrounded by non-polar alkyl groups are water- repelling in nature. They have in general high thermal stability, high dielectric strength and resistance to oxidation and chemicals. They are used as sealants greases, electrical insulators and for water-proofing of fabrics.
A large number of silicates minerals exist in nature. Some of examples are feldspar, zeolites, micas and asbestos. The basic structural unit of silicates is SiO4-4 (Fig.) in which silicon atom is bonded to four oxygen atoms in tetrahedron fashion. In silicates either the discrete unit is present or a present or a number of such units are joined together via corners by sharing 1,2,3 or 4 oxygen atoms per silicate units.
When silicate units are linked together, they form chain, ring, sheet or three-dimensional charged metal ions. If all the four corners are shared with other tetrahedral units, three-dimensional network is formed. Two important man made silicate ire glass and cement.
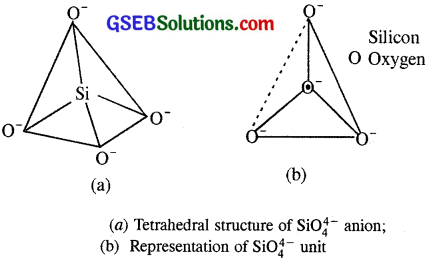
Zeolites : If aluminium atoms replace few silicon atoms in three-dimensional network of silicon dioxide, overall structure known as aluminosilicate, acquires a negative charge. Cations such as Na+, K+ or Ca2+ balance the negative charge.
Examples are feldspar and zeolites. Zeolites are widely used as a catalyst in petrochemical industries for cracking of hydrocarbons and isomerisation, example ZSM-5 (A type of zeolite) used to convert alcohols directly into gasoline. Hydrated zeolites are used as ion exchangers in softening of hard water.
![]()
Question 3.
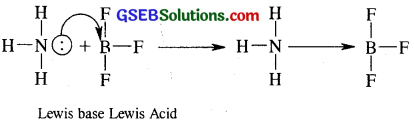
Why does boron triflouride behave as a Lewis acid?
Answer:
The Lewis structure of BF3 is 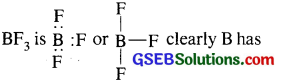
not been able to complete its octet [It has only 6 electrons around it]. Therefore BF3 is a electron deficient compound. All such electron- deficient compounds are electron pair acceptors and so are Lewis acids.
Question 4.
Consider the compounds, BCl3 and CCl4. How will they behave with water? Justify.
Answer:
BCl3 is hydrolysed by water completely to give boric acid and hydrochloric acid.
BCl3 + 3H2O → H3BO2 + 3HCl
Due to formation of HCl as a result of hydrolysis, BCl3 fumes in air.
CCl4, on the other hand, cannot undergo hydrolysis as it cannot expand its coordination number beyond four because of the absence of (i-orbitals. CCl4 is insoluble in water.
Question 5.
Is boric acid a protic acid? Explain.
Answer:
It is not a protonic acid, but acts as a Lewis acid by accepting electrons from a hydroxyl ion.
B(OH)3 + 2HOH → [B(OH)4]– + H3O+
Question 6.
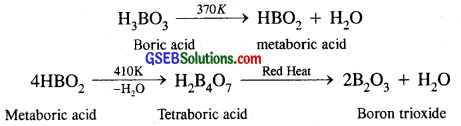
Explain what happens when boric acid is heated?
Answer:
Boric acid, on heating, loses water in three different stages at different temperatures ultimately giving boron trioxide.
Question 7.
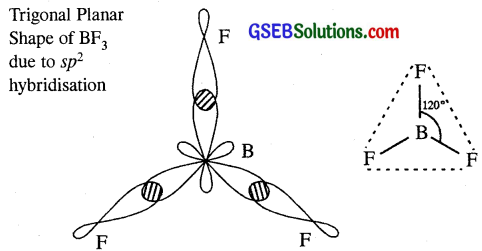
Describe the shapes of BF3 and BH–4. Assign the hybridisation of boron in these species.
Answer:
In BF3, boron undergoes sp² hybridisation. The electronic config. of B = 1s², 2s² 2P1-x
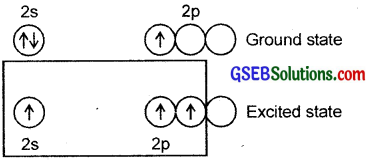
One 2s and two 2p atomic orbitals of B undergo sp² hybridisation giving three sp² hybrid orbitals.
BH–4 In BH–4 , boron shows sp² hybridisation and the shape of BH–4 is tetrahedral
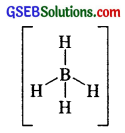
Tetrahedral shape of BH–4 due to sp3 hybridisation of Boron
![]()
Question 8.
Write reactions to justify amphoteric nature of aluminium.
Answer:
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character.
1. Aluminium dissolves in dilute HCl and liberates dihydrogen.
2Al(s) + 6HCl(aq) → 2Al3+ (aq) + 6Cl–(aq) + 3H2(g)
2. Aluminium also reacts with aqueous alkali and liberates dihydrogen.
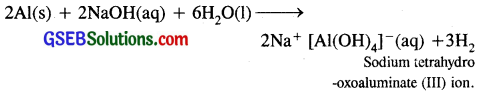
Question 9.
What are electron-deficient compounds? Are BCl3 and SiCl4 electron-deficient species? Explain.
Answer:
In trivalent state, the number of electrons around the central atom B in BCl is only six

Such electron-deficient molecules have a tendency to accept a pair of electrons to achieve stable electronic configuration and thus, behave as Lewis acids. Thus BCl3 is electron-deficient and is so a Lewis acid.
On the other hand SiCl4 is tetrahedral in shape due to sp3 hybridisation and in it silicon has already completed its octet.
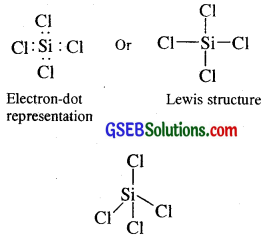
Tetrahedral shape of SiCl4 due to sp3 hybridisation Therefore SiCl4 is not an electron-deficient compound.
Question 10.
Write the resonance structures of CO2-3 and HCO–3.
Answer:
Resonance structures of CO2-3
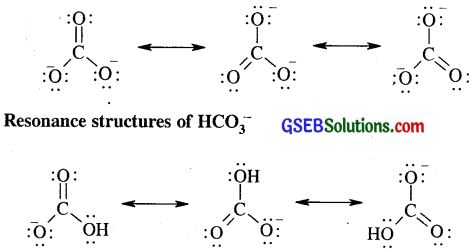
Question 11.
What is the state of hybridisation of carbon in
(a) CO2-3
(b) diamond
(c) graphite ?
Answer:
(a) Carbon shows sp² hybridisation in CO2-3 ion
(b) Carbon shows sp3 hybridisation in diamond
(c) Carbon shows sp² hybridisation in graphite.
Question 12.
Explain the difference in the properties of diamond and graphite on basis of their structures.
Answer:
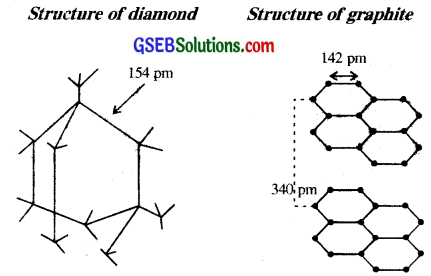
In diamond C is sp3 hybridised. Each C is tetrahedrally linked to four neighbouring carbon atoms through strong C-C sp3-sp3 N a-bonds. The structure is highly rigid and this network extends to three dimensions. Whereas in graphite C is sp² sheet like (layered) structure consisting of a number of benzene rings fused together.
The various sheets or layers are held together by weak van der Waals forces. Based on the above structural differences between diamond and graphite can be summed up as follows :
Diamond:
- Purity : It is the purest form of carbon.
- Bond length : Due to sp3 hybridisation C-C bond length is l54 pm
- Hardness : It is the hardest substance known with high density and high melting point.
- Conductivity : Since there are no free electrons, diamond is a bad conductor of electricity.
- Transparency: Because of high refractive index (2.5) it can reflect and refract light. It is a transparent substance
Graphite:
- Like diamond, graphite is also purest form of carbon.
- Bond lengths of C-C bonds in graphite due to sp² hybridisation is 142 pm.
- Since the successive layers can slip over each other, graphite is soft. It is a good lubricant.
- The 4th electron of each C atom is free. Therefore graphite is a good conductor of electricity and heat.
- Opaqueness : Unlike diamond, graphite is a black substance and possesses a metallic lustre.
![]()
Question 13.
Rationalise the given statements and give chemical reactions:
- Lead (II) chloride reacts with Cl2 to give PbCl4.
- Lead (IV) chloride is highly unstable towards heat.
- Lead is known not to form an iodide Pbl4.
Answer:
1. Lead (II) chloride PbCl2 does not react with Cl2 to form PbCl4. PbCl4 is a strong oxidising agent. PbCl4 decomposes on heating to give PbCl2 and Cl2.
![]()
2. Lead (II) chloride PbCl2 is thermally and chemically more stable than lead (IV) chloride PbCl4.
3. Pbl4 does not exist because Pb-I bond initially formed during the reaction of Pb with I2 does not release enough energy to unpair 6s² electrons and excite one of them to higher orbital to have 4 unpaired electrons around it,
Question 14.
Suggest reasons why the B-F bond lengths in BF3 (130 pm) and BF–4 (143 pm) differ?
Answer:
Boron in BF3 is sp² hybridised and it gives trigonal planar structure. Due to sp² hybridisation B-F bond in BF is shorter (130 pm). Boron in BF–4 is sp² hybridised and it gives tetrahedral structure. Due to sp³ hybridisation B-F bond in BF–4 is longer (143pm).
Question 15.
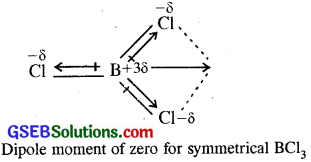
If B-Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
Answer:
BCl3 molecule due to sp² hybridisation of boron is trigonal planar molecule and has a symmetrical shape. Although individual B- Cl bond, has dipole, the molecule BCl3 has a zero dipole moment. This is due to the fact that individual dipole moments cancel out on account of the symmetry of the molecule as shown, below :
Question 16.
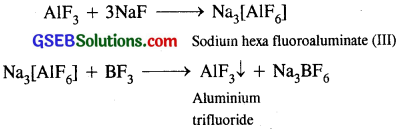
Aluminium trifluoride is insoluble in anhydrous HF but dissolves on addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous BF3 is bubbled through. Give reasons.
Answer:
AlF3 is insoluble in an hydrous HF, but dissolves on addition of NaF due to die formation of Na3AlF6
Question 17.
Suggest a reason as to why CO is poisonous.
Answer:
The poisonous nature of carbon monoxide (CO) is due to its ability to form a complex with haemoglobin which is about 300 times more stable than the oxygen-haemoglobin complex. This prevents haemoglobin in the red blood corpuscles from carrying oxygen round the body and ultimately resulting in death.
Haemoglobin + CO → Carboxyhaemoglobin.
![]()
Question 18.
How is excessive content of CO2 responsible for global warming?
Answer:
CO2 gas as such is not poisonous. CO2 which is normally present to the extent of 0.03 % by volume in the atmosphere, is removed from it by the process known as photosynthesis. It is the process by which green plants convert atmospheric CO2 into carbohydrates such as glucose.
But the increases in the combustion of fossil fuels and decomposition of lime stone for cement manufacture in recent years seem to increase the CO2 content of the atmosphere. This may lead to green house effect and thus, raise the temperature of the atmosphere which might have serious consequences.
Question 19.
Explain structures of diborane and boric acid.
Answer:
Structure of diborane : The four terminal hydrogen atoms and the two boron atoms lie in one plane. Above and below this plane, there are two bridging hydrogen atoms. The four terminal B-H bonds are regular two centre-two electron bonds while the two bridge (B-H-B) bonds are different and can be described in terms of three centre-two electron bonds as shown in the figure below.
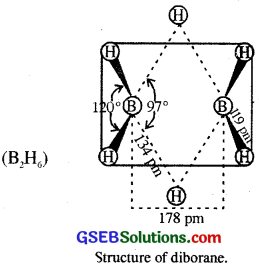
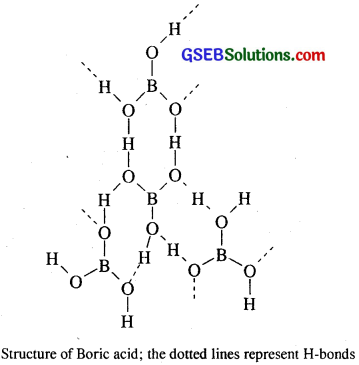
Structure of boric acid (H3BO3) : Orthoboric acid or simply boric acid (H3BO3) has a layer structure in which BO3 units are joined by hydrogen bonds.
Question 20.
What happens when
(a) Borax is heated strongly
(b) Boric acid is added to water
(c) Aluminium is treated with dilute NaOH
(d) BF3 is treated with ammonia?
Answer:
(a) On heating borax it first loses water molecules and swells up. On further heating, it turns into a transparent liquid which solidifies into glass like material known as borax bead.

(b) Boric acid is sparingly soluble in cold water, but fairly soluble in hot water. Boric acid behaves as a weak monobasic acid (Ka = 1.0 x 10-9). It does not act as a proton-donor, i.e., protonic acid, but behaves as a LEWIS-ACID, i.e., it accepts a pair of electrons from OH– ion.
B(OH)3 + 2H-OH → [B(OH)4]– + H3O+
(c) Aluminium is treated with dilute NaOH: It liberates H2
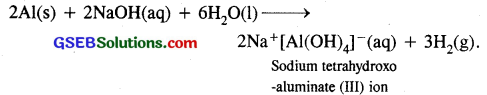
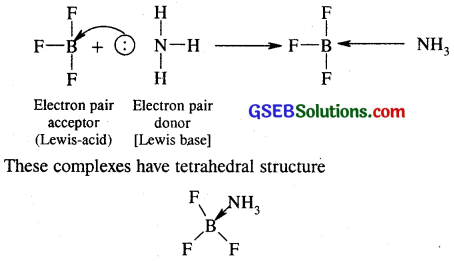
(d) BF3 is treated with amonia. BF3 being a strong Lewis acid reacts with NH3 to form adduct or complex
Question 21.
Explain the following reactions
(a) Silicon is heated with methyl chloride at high temperature in the presence of copper;
(b) Silicon dioxide is treated with hydrogen fluoride;
(c) CO is heated with ZnO;
(d) Hydrated alumina is treated with aqueous NaOH solution.
Answer:
(a) When silicon is heated with methyl chloride at high temperature in the presence of copper as catalyst, various types of methyl substituted chlorosilanes of formula MeSiCl3, Me2SiCl2, Me2SiCl with small amount of Me3Si are formed.

Hydrolysis of dimethyl dichloro silane (CH3)2SiCl2 yields straight chain polymers.
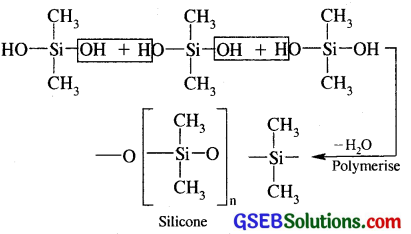
(b) When silicon dioxide is treated with hydrogen fluoride (HF), SiF4 (Silicon tetra fluoride) is formed
SiO2 + 4HF → SiF4 + 2H2O
(c) When CO is heated with zinc oxide (ZnO), it is reduced to zinc by carbonmonoxide (CO)
ZnO + CO → Zn + CO2
(d) When hydrated alumina is treated with aqueous NaOH solution, the following reaction takes place and sodium meta aluminate is formed.
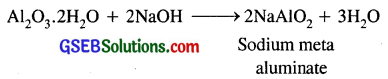
Question 22.
Give reasons :
(i) Cone. HNO3 can be transported in aluminium container.
(ii) A mixture of dilute NaOH and aluminium pieces is used to open drain.
(iii) Graphite is used as lubricant.
(iv) Diamond is used as an abrasive
(v) Aluminium alloys are used to make aircraft body.
(vi) Aluminium utensils should not be kept in water overnight.
(vii) Aluminium wire is used to make transmission cables.
Answer:
(i) Cone. HNO3 can be transported in aluminium container
because concentrated nitric acid (HNO3) renders aluminium passive by forming a protective oxide layer on the surface.
2HNO3 → H2O + 2NO2 + O
2Al + 3O → Al2O3 (passive protective layer)
(ii) A mixture of dil NaOH and aluminium pieces is used to open drain because of the liberation of H2 (gas)
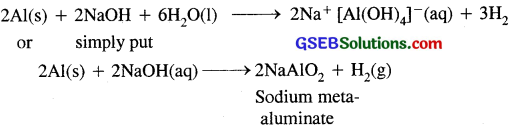
(iii) Graphite is used as a lubricant because its layers can slip over each other and therefore it is very soft and slippery. It is used as a dry lubricant in machines running at high temperature, where oil cannot be used as a lubricant.
(iv) Diamond is used as an abrasive. The rigid three-dimensional network of carbon atom extends in space in diamond through strong covalent C – C bonding. It is very difficult to break extended covalent bonding and therefore, diamond is the hardest substance on the earth. Therefore, it is used as an abrasive for sharpening hard tools.
(v) Aluminium alloys are used to make aircraft body, because its alloys with other metals makes it highly tensile and due to its light weight nature it can be given any shapes of pipe, tubes, rods, wires, plates or foils etc.
(vi) Aluminium utensils should not be kept in water overnight because in the presence of water and oxygen, a thin toxic layer of Al2O3 is formed on its surface.
(vii) Aluminium wire is used to make transmission wires because aluminium possesses high electrical conductivity. On a weight-to- weight basis the electrical conductivity of aluminium is twice that of copper.
![]()
Question 23.
Explain why is there a phenomenal decrease in ionisation enthalpy from carbon to silicon.
Answer:
Electronic conftg. of C is = 1s², 2s² 2p²
Electronic conftg. of Si = 1s², 2s²2p6, 3s²3p²
As we proceed from C to Si in group l4, there is a sharp increase in covalent radius from 77 pm for P to 118 for silicon. Due to the increase in the size of the atom, there is a sharp fall in the value of ionisation enthalpy from carbon to silicon.
Question 24.
How would you explain the lower atomic radius of Ga as compared to Al?
Answer:
The electronic conftg. of Al = [Ne] 3s²3p1]
Ga = [Ar] 3d104s²4py
Gallium has 10 additional d-electrons in its inner shell or core of its atom. The presence of additional 10 – d electrons offer only poor screening effect for the outer electrons from the increased nuclear charge in gallium (Ga). Consequently, the atomic radius of gallium (135 pm) is less than that of aluminium (143 pm).
Question 25.
What are allotropes? Sketch the structure of two allotropes of carbon namely diamond and graphite.
or
What is the impact of structure on physical properties of two allotropes?
Answer:
When an element exists in two or more different physical forms having similar chemical properties, but different physical properties it is said to be present in two or more allotropes or allotropic modifications. Carbon exists in two allotropic forms as diamond and graphite.
Whereas C shows sp³ hybridisation in diamond it shows only sp² hybridisation in graphite. Due to the absence of any free electron in diamond, it is a non-conductor of electric current, whereas the presence of one free electron in graphite makes it a conductor of electric current.
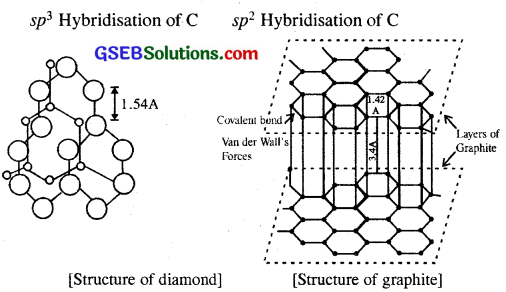
Impact of the two structures of diamond and graphite on physical properties of two allotropes.
- Diamond is the hardest substance known and can cut glass whereas graphite is so soft that it marks paper.
- Diamond is a transparent substance, whereas graphite is opaque.
- Diamond cannot conduct electricity, whereas graphite can.
- Specific gravity of diamond is 3.5, whereas it 2.5 for graphite.
Question 26.
(a) Classify following oxides as neutral, acidic, basic or amphoteric : CO, B2O3, SiO2, CO2, Al2O3, PbO2, TI2O3.
(b) Write suitable chemical equations to show their nature.
Answer:
(a)
- CO is neutral
- B2O3, SiO2, CO2 are acidic oxides
- PbO2, Al2O3 are amphoteric oxides
- TI2O3 is basic oxide.
(b)
- B2O3 + CuO → Cu(BO2)2
- SiO2 + 2NaOH → Na2SiO3 + H2O
- CO2 + H2O → H2CO3
- PbO2 + 4HCl → PbCl4 + 2H2O
- PbO2 + 2NaOH → Na2PbO3 + H2O
- Al2O3 + 6HCl → 2AlCl3 + 3H2O
- Al2O3 + 2NaOH → 2NaAlO2+ H2O
- Tl2O3 + 3H2SO4 → Tl2(SO4)3 + 3H2O
![]()
Question 27.
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group-I metals. Support this statement by giving some evidences.
Answer:
Like aluminium compounds thallium also shows an oxidation state of +3 in compounds like Tl2O3, because both Al and Tl belong to group 13 and have ns²np1 electronic config. in their valence shells.
Like alkali metals thallium shows an oxidation state of +1 in its halides.
2Na + Cl2 → 2NaCl
2Tl + Cl2 → 2TlCl
It is due to the fact that heavy elements like Tl show inert pair effect. Thus it prefers to show an ox. state of + 1 like alkali-metals.
Question 28.
When metal X is treated with sodium hydroxide, a wsate precipitate (A) is obtained, which is soluble in excess of NaOH to give soluble complex (B). Compound (A) is soluble in dilute HCl to form compound (C). The compound (A) when heated strongly gives (D), which is used to extract metal. Identify (X), (A), (B), (C) and (D). Write suitable equations to support their identities.
Answer:
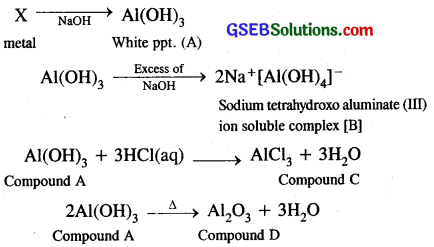
∴ X is metal aluminium
Compound A is Al(OH)3
Soluble complex is Na+[Al(OH)4]–
Compound C is AlCl3
Compound D is Al2O3
Question 29.
What do you understand by
(a) Inert pair effect
(b) allotropy
(c) catenation.
Answer:
(a) Inert pair effect: The heavier elements of groups 13, 14, 15 show an oxidation state two less than the number of valence electrons they possess.
Group 1 : Electronic config. ns²np1
Group 14 : Electronic config. ns²np²
Group 15 : Electronic config. ns²np3
The last element of these groups Tl, Pb, Bi prefer to show oxidation states of +1, +2, +3 in preference over +3, +4, +5 which the elements of these groups normally exhibit. This is due to the fact that as we go down in these groups, the 2 electrons of ns² prefer to remain paired and do not participate in bond formation. Such a pair of electrons is called inert pair and the effect is called INERT PAIR EFFECT.
(b) Allotropy : When an element exists in two or more different forms, it is said to exihibit allotropy and the different forms are called allotropes. For example, phosphorus occurs in three allotropic forms, i.e., white, red and black. Sulphur shows two different solid forms : Sulphur rhombic and sulphur monoclinic.
(c) Catenation : Property of self-linking of atoms of the same element through strong covalent bonds is called catenation, example C-C bonds in hydrocarbons. It is due to strong C-C covalent bonds that catenation is exihibited.
![]()
Question 30.
A certain salt X, gives the following results.
(i) Its aqueous solution is alkaline to litmus.
(ii) It swells up to a glassy material Y on strong heating.
(iii) When cone. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out
Write equations for all the above reactions and identify X, Y and Z.
Answer:
The salt X is BORAX (Na2B4O7)
(i) Its aqueous solution in water is alkaline due to hydrolysis. Borax is the salt of a strong base NaOH and a weak acid H2B4O7.
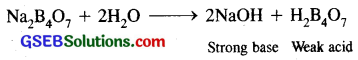
Such a solution is alkaline to litmus.
(ii) It swells up to a glassy material Y on strong heating
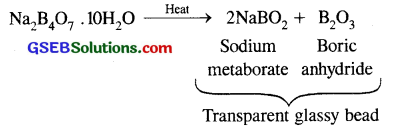
Borax loses its water of crystallisation on heating and swells up to form a puffy mass of formula N2B4O7.
(iii) When cone. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out.
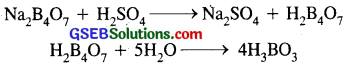
∴ Z is boric acid (H3BO3) whose white crystals separate out when cone. H2SO4 is added to the hot solution of X.
∴ X is borax (Na2B4O7) Y is anhydrous Na2B4O7, Z is boric acid H3BO3.
Question 31.
Write balanced equations for :
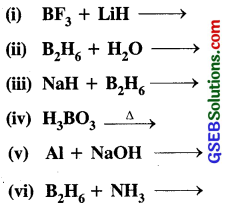
Answer:
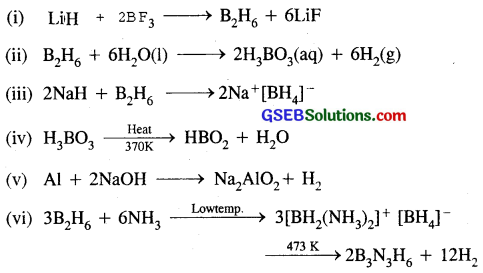
Question 32.
Give one method for industrial preparation and one for laboratory preparation of CO and CO2 each.
Answer:
CO : On a small scale pure CO is prepared by dehydration of formic acid (HCOOH) with concentrated H2SO4 at 373K
![]()
On a commercial scale CO is prepared by the passage of steam over hot coke
C(s) + H2O(g) → CO(g) + H2(g)
CO2 : In the laboratory CO2 is prepared by the action of dilute HCl acid on CaCO3.
CaCO3 + 2HCl → CaCl2 + H2O + CO2
On a commercial scale it is prepared by heating lime stone.

Question 33.
An aqueous solution of _________.
(a) neutral
(c) basic
Answer:
(c) It is basic.
![]()
Question 34.
Boric acid is polymeric due to _________.
(a) Its acidic nature
(b) The presence of hydrogen bonds
(c) Its monobasic nature
(d) Its geometry.
Answer:
(b) The presence of hydrogen bonds.
Question 35.
The type of hybridisation of boron in diborane is _________.
(a) sp
(b) sp²
(c) sp³
(d) dsp².
Answer:
(c) sp³ hybridisation of B in diborane B2H6.
Question 36.
Thermodynamically the most stable form of carbon is _________.
(a) Diamond
(b) Graphite
(c) Fullerenes
(d) Coal.
Answer:
(b) Graphite is thermodynamically most stable allotrope of carbon.
Question 37.
Elements of group 14 _________.
(a) exhibit oxidation state of + 4 only .
(b) exhibit oxidation state of + 2 and + 4
(c) form M2- and M2+ ion
(d) form M2+ and M4+ ions.
Answer:
(b) Elements of group 14 exhibit oxidation states of + 2 and + 4.
![]()
Question 38.
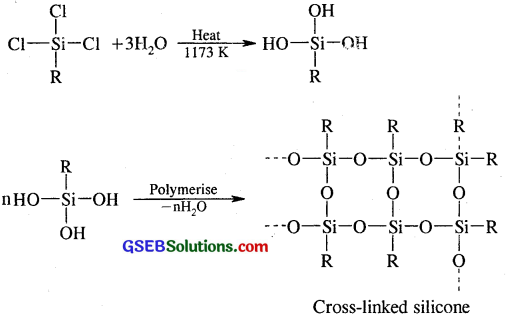
If the starting material for the manufacture of silicones is RSiCl3, write the structure of the product formed.
Answer:
If the starting material for the manufacture of silicones is RSiCl3, cross-linked polymers are obtained as detailed below