Gujarat Board GSEB Textbook Solutions Class 7 Science Chapter 5 Acids, Bases and Salts Textbook Questions and Answers, Notes Pdf.
Gujarat Board Textbook Solutions Class 7 Science Chapter 5 Acids, Bases and Salts
Gujarat Board Class 7 Science Acids, Bases and Salts Textbook Questions and Answers
![]()
Question 1.
State differences between acids and bases.
Answer:
Acids:
- Acids are sour to taste.
- Acid turns blue litmus to red.
- Acid is a sub-stance which contains hydrogen ion (H+).
Bases:
- Bases are bitter to taste.
- Base turns red litmus to blue.
- Bases are substances which contain hydroxylion (OH–).
Question 2.
Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. What is its nature?
Answer:
Ammonia has basic nature.
![]()
Question 3.
Name the source from which litmus solution is obtained. What is the use of this solution?
Answer:
Litmus solution is extracted from lichens. It is used to determine whether the given solution is acidic or basic.
Question 4.
Is the distilled water acidic/basic/ neutral? How would you verify it?
Answer:
Distilled water is neutral. We can verify it by showing that neither blue nor red litmus paper changes its colour when dipped in it.
Question 5.
Describe the process of neutralisation with the help of an example.
Answer:
The reaction between an acid and a base is known as neutralisation. Salt and water are produced in this process with the evolution of heat. Antacids like milk of magnesia (magnesium hydroxide), baking soda, etc. which contain a base are used for reducing acidity in stomach when excessive acid is released by glands.
![]()
Question 6.
Mark ‘T’ if the statement is true and ‘F’ if it is false:
(i) Nitric acid turns red litmus blue.
(ii) Sodium hydroxide turns blue litmus red.
(iii) Sodium hydroxide and hydrochloric acid neutralise each other and form salt and water.
(iv) Indicator is a substance which shows different colours in acidic and basic solutions.
(v) Tooth decay is caused by the presence of a base.
Answer:
(i) False
(ii) False
(iii) True
(iv) True
(v) False
Question 7.
Dorji has a few bottles of soft drink in his restaurant. But, unfortunately, these are not labelled. He has to serve the drinks on the demand of customers. One customer wants acidic drink, another wants basic and third one wants neutral drink. How will Dorji decide which drink is to be served to whom?
Answer:
Dorji can decide with the help of litmus paper:
- The drink which would turn a red litmus blue would be basic.
- If the drink turns a blue litmus to red, it would be acidic.
- The drink which would not affect both red and blue litmus would be neutral.
Question 8.
Explain why:
(a) An antacid tablet is taken when you suffer from acidity.
(b) Calamine solution is applied on the skin when an ant bites.
(c) Factory waste is neutralised before disposing it into the water bodies.
Answer:
(a) We take an antacid such as milk of magnesia to neutralise the excessive acid released in stomach.
(b) Ant injects an acidic liquid (Formic acid) into the skin on biting which causes inflammation to the skin. The effect of the acid can be neutralised by rubbing. Calamine solution which contains zinc carbonate which is very weak base and causes no harm to the skin.
(c) The wastes of factories contain acids. If acids are disposed off in the water body, the acids will harm the organisms. So factory wastes are neutralised by adding basic substances.
![]()
Question 9.
Three liquids are given to you. One is hydrochloric acid, another is sodium hydroxide and third is a sugar solution. How will you identify them? You have only turmeric indicator.
Answer:
Name of the substances:
- Hydrochloric acid
- Sodium hydroxide
- Sugar solution
Effect on turmeric indicator:
- Yellow to blue
- Yellow to red
- No change
Question 10.
Blue litmus paper is dipped in a solution. It remains blue. What is the nature of the solution? Explain.
Answer:
(i) It can be identified on the basis of the following observations : Bases change the colour of Red litmus paper to blue. As the colour of blue litmus paper is not affected, the solution must be basic.
(ii) If the solution is neutral, even then colour of litmus will not change.
Question 11.
Consider the following statements:
(a) Both acids and bases change colour of all indicators.
(b) If an indicator gives a colour change with an acid, it does not give a change with a base.
(c) If an indicator changes colour with a base, it does not change colour with an acid.
(d) Change of colour in an acid and a base depends on the type of the indicator. Which of these statements are correct?
(i) All four
(ii) (a) and (d)
(iii) (b) and (c)
(iv) only (d)
Answer:
(ii) (a) and (d)
Extended Learning – Activities And Projects
Question 1.
Using the knowledge of acids and bases, write a secret message with the help of baking soda and beetroot. Explain how it works.
(Hint: Prepare baking soda solution in water. Use this solution to write the message on a sheet of white paper with a cotton bud. Rub a slice of fresh beetroot over the message.)
Answer:
Prepare baking soda solution in water. Use this solution to write the message on a sheet of white paper with a cotton bud. We observe that we cannot read the message and cannot see it. Now rub a slice of fresh beetroot over the message then we can see it and read it. It becomes visible but before the rubbing of beetroot it was hidden. Therefore, it is a secret message. Actually, baking soda solution is basic in nature. It reacts with beetroot juice (indicator) and words become visible.
![]()
Question 2.
Prepare red cabbage juice by boiling a piece of red cabbage in water. Use it as an indicator and test the acidic and basic solutions with it. Present your observations in the form of a table.
Answer:
We prepare the indicator by boiling a red cabbage piece. Now we observe its effect on following substances.
Answer:
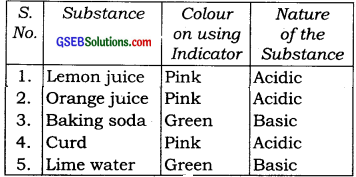
Question 3.
Bring the soil sample of your area, find out if it is acidic, basic or neutral. Discuss with farmers if they treat the soil in any manner.
Answer:
Take the samples of soil from a field of your areas. Mix the soil with water in a beaker. Stir well the mixture. Filter the mixture and take the filtrate in ‘ another beaker. Test it with litmus paper. If it converts blue litmus into red then its nature is acidic. If it converts red litmus into blue then it is basic. If there is no effect on litmus paper then it is neutral. This is the best way and easiest way to test the nature of soil by the farmers.
![]()
Question 4.
Visit a doctor. Find out the medicines, he prescribes to treat acidity. Ask him how acidity can be prevented.
Answer:
The medicines prescribed by the doctors are basic in nature and are called antacids. These medicines neutrilise the effect of acids.