Gujarat Board GSEB Textbook Solutions Class 8 Science Chapter 14 Chemical Effects of Electric Current Textbook Questions and Answers, Notes Pdf.
Gujarat Board Textbook Solutions Class 8 Science Chapter 14 Chemical Effects of Electric Current
Gujarat Board Class 8 Science Chemical Effects of Electric Current Textbook Questions and Answers
Question 1.
Fill in the blanks:
(а) Most liquids that conduct electricity are solutions of …………… and ………….
(b) The passage of an electric current through a solution causes effects ………….
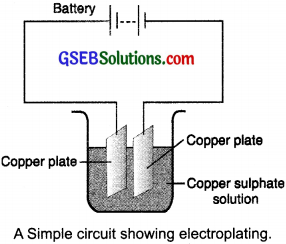
(c) If you pass current through copper sulfate solution, copper gets deposited on the plate connected to the ………… terminal of the battery …………..
(d) The process of depositing a layer of any desired metal on another material, by means of electricity, is called ………..
Answer:
(a) acids, bases, salts
(b) chemical
(c) negative
(d) electroplating.
![]()
Question 2.
When the free ends of a tester are dipped into a solution, the magnetic needle shows deflection. Can you explain the reason?
Answer:
Yes, we can explain the reason. The magnetic needle will show deflection when the circuit is complete. The deflection of the magnetic needle shows that the solution is a good conductor.
Question 3.
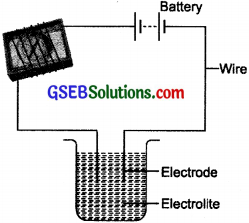
Name three liquids, which when tested in the manner shown in Fig. 14.1 may cause the magnetic needle to deflect.
Answer:
Tap water, hydrochloric acid, sodium hydroxide.
Question 4.
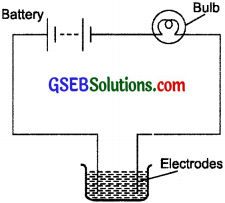
The bulb does not glow in the set up shown in Fig. 14.2. List the possible reasons. Explain your answer.
Answer:
The possible reasons may be:
- The connections of the circuit may be loose.
- The bulb may be fused.
- Cells may be used up.
- The liquid may be a poor conductor.
Question 5.
A tester is used to check the conduction of electricity through two liquids, labelled A and B. It is found that the bulb of the tester glows brightly for liquid A while it glows very dimly for liquid B. You would conclude that:
- liquid A is a better conductor than liquid B.
- liquid B is a better conductor than liquid A.
- both liquids are equally conducting.
- conducting properties of liquid cannot be compared in this manner.
Answer:
1. liquid A is a better conductor than liquid B
![]()
Question 6.
Does pure water conduct electricity? If not what can we do to make it conducting?
Answer:
No, pure water does not conduct electricity. Pure water can be made conducting by dissolving salt in it.
Question 7.
In case of a fire, before the firemen use the water hoses, they shut off the main electrical supply for the area. Explain why they do this.
Answer:
The water used in water hoses is a good conductor of electricity. Firemen shut off the main electrical supply, because if the supply of electricity continues there may be a high risk of electrocution due to water.
Question 8.
A child staying in a coastal region test the drinking water and also the seawater with his tester. He finds that the compass needle deflects more in the case of seawater. Can you explain the reason?
Answer:
Seawater contains more amount of salt than drinking water. So the seawater is a better conductor of electricity. This is the reason that the compass needle deflects more in the case of seawater.
![]()
Question 9.
Is it safe for the electrician to carry out electrical repairs outdoors during heavy downpour? Explain.
Answer:
No, it is not safe for the electrician to carry out electrical repairs during a heavy downpour. It is because during heavy downpour there is a higher risk of electrocution.
Question 10.
Paheli had heard that rainwater is as good as distilled water. So she collected some rainwater in a clean glass tumbler and tested it using a tester. To her surprise, she found that the compass needle showed deflection. What could be the reasons?
Answer:
Rainwater is like distilled water but some impurities are mixed in from the atmosphere. These impurities make the rainwater conducting. This could be the reason for the deflection of the compass needle.
Question 11.
Prepare a list of objects around you that are electroplated.
Answer:
Pots of metals, bath taps, ornaments, rims of vehicles, the handlebar of cycles and motorcycles, kitchen gas burner, the bottom of cooking utensils, handles of doors, tin cans are some objects around us that are electroplated.
![]()
Question 12.
The process that you saw in Activity 14.7 is used for the purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from the impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of the battery and why?
Answer:
The rod of the copper plate should be connected to the positive terminal of the battery. Because when an electric current is passed through the copper sulfate solution, it dissociates into copper and sulfate. The free copper drawn to the negative terminal of the battery gets deposited on it. On the other hand, the loss of copper from the solution would be regained from the impure copper rod which is connected to the positive terminal of the battery.